Nitinol is well suited to the design constraints imposed by medical device applications, however, only modest progress has so far been made regarding long term predictions of the structural integrity of such components.
Challenges in predicting long term reliability of superelastic NiTi medical components include the consequences of manufacturing tolerances, uncertainty with regard to in vivo loading and the inherent complexity of superelastic material behavior. In this article, we focus on the latter challenge and discuss ongoing research to characterize the effects of phase transformation and accurately obtain calibrated material constitutive relations for polycrystalline Nitinol materials.
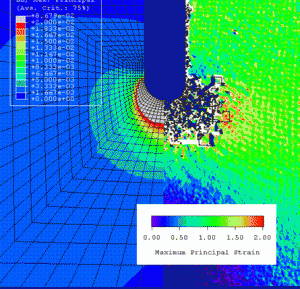
Our approach involves a combination of full-field strain measurements using phase shifted Moire interferometry and the development of improved finite element methodology to account for various material behaviors and simulated conditions. As you can see in our presentation, phase shifted Moire interferometry provides unprecedented accuracy in the measurement of strains and displacements in the vicinity of stress risers such as notches and can be used to guide and verify finite element models for implantable superelastic devices.
We originally presented this work at the American Society for Materials (ASM) conference in 2004.
Download the Abstract and presentation